Every year about 500,000 people become disabled as a result of spinal cord injuries (SCI). The communication lines between the brain and spinal cord below the injury are cut or dramatically diminished, depending on the severity of the event, which leads to a range of motor disabilities.
It is possible to access the surviving circuits and pathways to alleviate these deficits via epidural electrical stimulation (EES). Walking requires the activation of spatially distributed spinal motor circuits following precise temporal sequences that are continuously adjusted through sensory feedback. Therefore, current neuromodulation therapies – which deliver stimulation to restricted spinal cord locations and remain constant throughout gait execution – are not optimal. In the present work, we argued that targeted stimulation in space and time of the spinal cord, matching the natural dynamics of spinal motor circuit activation, can restore walking and improve motor control after spinal cord injury.
We first conducted anatomical and functional experiments to visualize the spatiotemporal pattern of hindlimb motoneuron activation in intact rats (Figure 1a). We found that walking involves the alternating activation of spatially restricted hotspots underlying extensor versus flexor muscle synergies. We then developed neuromodulation strategies that specifically target proprioceptive feedback circuits in the dorsal roots in order to access these hotspots. Computer simulations determined the optimal electrode locations to recruit specific subsets of dorsal roots. These results steered the design of spatially selective spinal implants and real–time control software to modulate extensor versus flexor muscle synergies with precise temporal resolution adjusted through movement feedback. This conceptually new stimulation strategy reinforced extension versus flexion components for each hindlimb independently and improved a range of important gait features after complete SCI (see Figure 1b).
We considered that spinal implants designed to activate the proprioceptive afferents projecting to the identified flexor and extensor hot spots would engage muscle synergies encoders related to extension and flexion. Our results showed that tailored spinal implants targeting specific subset of dorsal roots with electrodes enabled a gradual control over the degree of flexion and extension on the left and right hindlimbs. Although challenges lie ahead, we believe that spatiotemporal neuromodulation of the spinal cord will become a viable way to accelerate and augment functional recovery in humans with SCI.
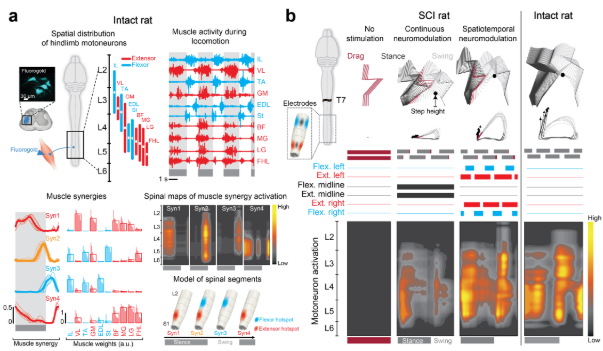
Figure 1: Spatiotemporal neuromodulation reproduces the natural pattern of motoneuron activation. From left to right and top to down. (a) Tracer injections into muscles spanning each hindlimb joint, to visualize the spatial location of hindlimb motoneurons. We decomposed the muscle activity during locomotion recorded in all the traced muscles into functional models (muscle synergies). To link muscle synergies to the activation of the burst (‘hotspot’) of motoneuron activity, we extracted the spinal map for each synergy independently. A model of spinal segments showing the temporal sequence underlying the recruitment of muscle synergies and the corresponding activation of extensor and flexor hot spots. (b) Rats received complete SCI at T7 and a spinal implant with conventional midline electrodes (black) and spatially selective lateral electrodes (blue and red). Locomotion in rats on a treadmill without stimulation and with continuous neuromodulation applied over the midline of lumbar and sacral segments (black electrodes) and during spatiotemporal neuromodulation (blue and red electrodes). On the right the results for an intact rat is showed.
Publication
Wenger N, Moraud EM, Gandar J, Musienko P, Capogrosso M, Baud L, Le Goff CG, Barraud Q, Pavlova N, Dominici N, Minev IR, Asboth L, Hirsch A, Duis S, Kreider J, Mortera A, Haverbeck O, Kraus S, Schmitz F, DiGiovanna J, van den Brand R, Bloch J, Detemple P, Lacour SP, Bézard E, Micera S, Courtine G (2016). “Spatiotemporal neuromodulation therapies engaging muscle synergies improve motor control after spinal cord injury.” Nature Medicine. Feb;22(2):138-45. http://www.nature.com/nm/journal/v22/n2/full/nm.4025.html
About the Author

Nadia Dominici
Associate Professor at the Department of Human Movement Sciences, Faculty of Behavioural and Movement Science, Vrije Universiteit of Amsterdam
Nadia Dominici, Associate Professor at the Department of Human Movement Sciences, Faculty of Behavioural and Movement Science at the Vrije Universiteit of Amsterdam, Research Institute MOVE, The Netherlands.
Nadia Dominici works on the interplay between brain and muscular activity underlying independent walking in children, as well as on the biomechanics of human locomotion. After a master diploma in Physics, she obtained a PhD in Neuroscience at the University of Rome “Tor Vergata”. She has held research positions at the Laboratory of Neuromotor Physiology of the Santa Lucia Foundation in Rome, where she focused on central pattern generation networks and on the development of locomotion in children, and at the University of Zürich, and EPFL in Lausanne, where she developed neurorehabilitation techniques to restore walking in animals after spinal cord injuries.
Copyright
© 2018 by the author. Except as otherwise noted, the ISPGR blog, including its text and figures, is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License. To view a copy of this license, visit https://creativecommons.org/licenses/by-sa/4.0/legalcode.
ISPGR blog (ISSN 2561-4703)
Are you interested in writing a blog post for the ISPGR website? If so, please email the ISGPR Secretariat with the following information:
- First and Last Name
- Institution/Affiliation
- Paper you will be referencing
